Employing such co-valorization tactics can indeed address current drawbacks of the OER by increasing revenues and the value proposition of the overall process. By coupling worthwhile oxidation reactions at the anode with cathode electrosynthesis, industries can leverage existing infrastructure for purification, distribution, and waste-management, and even produce feedstock chemicals required for parallel processes housed at the same facility. These reactions can tackle a range of objectives including raw chemical generation (e.g.H2O2), waste oxidation, and molecule upgrade. In light of these limitations, several OER alternatives have been proposed to decrease energy consumption and/or generate high-value products that can offset operation costs and improve sustainability. 
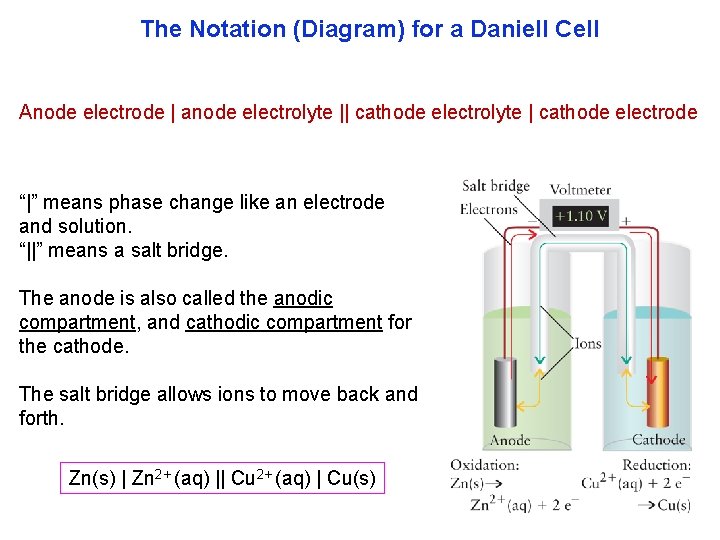
Nevertheless, the bulk of current electrolyzer designs rely on the traditional oxygen evolution reaction (OER) to extract electrons from water, which has been expensive, location limited, high-risk, and generates low value (O2) product. Anode: Pb(s)Pb2+(aq)+2e Ered0.13 V Cathode:Cl2(g)+2e2Cl(aq) Ered 1.36 V Since this is a voltaic cell, cell potentials must be assigned to give a positive Ecell. 
Due to the rapid decrease in the cost of renewable energy, it is now practical to design devices that use renewable electrons in electrochemical processes to drive the transformation of CO 2 and other waste feedstock (wastewater, food waste, biomass) into high-value products while also recovering important resources such as water, nutrients, and energy. To label the voltaic cell, start by writing out the half-reactions that occur at each half-cell and look up the standard reduction potentials. Understanding and advancing the water-energy-climate nexus is key to mitigating the immense threats of climate change and solving many of the related environmental issues we face today.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
